Choosing NEPA21 vs Viral Delivery in Brain Organoids
How labs decide based on organoid state, lumen access, cargo size, and mosaic vs stable expression.
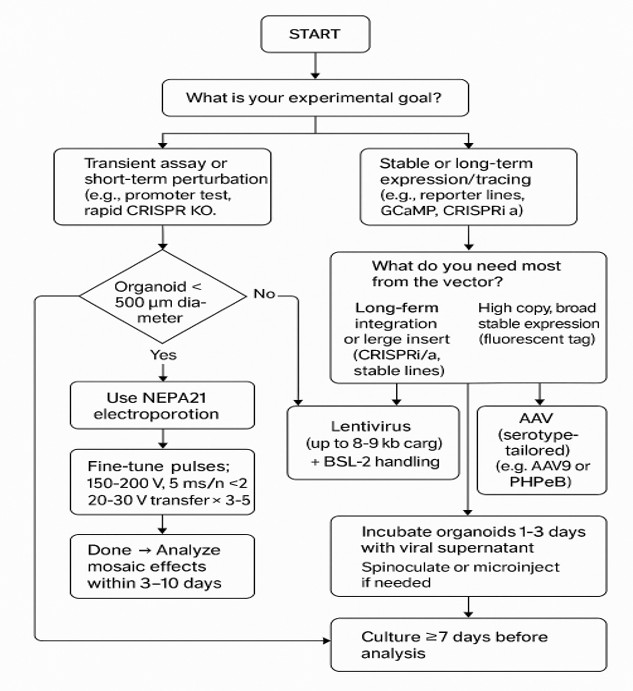
Brain organoids are thick, regionally patterned 3D tissues, so delivery strategy matters early. In practice, the best method usually depends on four things: organoid stage, access to ventricular-like lumens, cargo size, and whether you want mosaic or broad expression.
For many labs, the key choice is:
NEPA21 for fast, flexible, non-viral delivery
vs
Viral transduction for longer-term, more uniform expression
Navigation Shortcuts
Choose NEPA21 when you need:
- fast validation
- lumen-targeted delivery
- large cargo such as plasmids or CRISPR RNPs
- mosaic labelling or cell-autonomous phenotypes
Choose viral delivery when you need:
- stable expression over weeks to months
- broader labelling in thicker or more mature tissue
- long-term functional assays
- pooled or selection-based workflows
A practical workflow many labs use:
NEPA21 first for fast construct or guide validation → viral delivery later for stable assays
(Fast check)
| 1. | Do you need stable expression for weeks to months, lineage tracing, or pooled screens? |
| → Yes: Viral (usually lenti; AAV depending on target) → No / transient is fine: NEPA21 |
| 2. | Is the organoid early or small and lumen-accessible (< ~500 µm, thin ECM)? |
| → Yes: NEPA21 → it is thicker or more mature (> ~1 mm): Viral delivery is usually more practical |
| 3. | Is your cargo large or difficult to package into virus (CRISPR RNPs or big plasmids > 8 kb)? |
| → Yes: NEPA21(often simplest) → No: Either can work; decide based on duration and labelling pattern |
| 4. | Do you want mosaic, cell-autonomous phenotypes, or sparse labelling? |
| → Yes: NEPA21 → No: you want broad uniform labelling/expression: Viral |
NEPA21 vs Viral Delivery At a Glance
| Criterion | NEPA21 Electroporation | Viral Delivery (AAV / Lentivirus) |
|---|---|---|
| Expression duration | Transient, usually days to weeks | Longer-term, often weeks to months |
| Cargo flexibility | DNA, mRNA, and RNPs (large ok) | More packaging constraints; cDNA or CRISPRi/a typically ≤ 9 kb (AAV ~4.7 kb) |
| Best stage | Early organoids, rosettes, and lumen-accessible tissue | Mid-to-late organoids and thicker tissue |
| Labelling pattern | Mosaic, tuneable, and sparse-friendly | Broader, more uniform (MOI/ tropism dependent) |
| Throughput / cost | Low cost, fast turnaround (often same-day), no viral production | Slower setup and expression timeline |
| Biosafety | Minimal (non-viral) | Typically BSL-2 handling |
| Tissue access bias | Strong for lumen-facing and apical regions | Better suited to broader tissue exposure |
Common integration patterns labs use
1) In vitro IUE-style lumen electroporation (microinject → NEPA21)
- Use when: early cortical organoids or rosette structures where lumens are accessible.
- Why: supports rapid perturbation of apical progenitors and is especially useful for mosaic readouts.
2) Upstream engineering step before organoid formation
- Use when: you want to edit hPSCs or iPSCs first, then generate organoids from a stable engineered line.
- Why: efficient for knock-in / knock-out steps prior to organoid differentiation. It avoids viral workflows during line engineering and can simplify downstream differentiation studies.
3) Pilot-to-production pipeline (NEPA21 validation → viral for stable assays)
- Use when: the end goal is viral, (e.g. long-term calcium imaging), but you want to de-risk constructs/guides first.
- Why: reduces time lost on vector building and virus production for weak candidates.
4) Sparse labelling by design(NEPA21 mosaicism as a feature)
- Use when: you need single-cell morphology, migration analysis, or neighbour comparisons inside the same organoid.
- Why: mosaic delivery becomes an advantage rather than a limitation and enables cell-autonomous phenotype comparisons.
Why labs choose NEPA21 in brain organoids
NEPA21 is commonly chosen when labs need a delivery method that is fast, flexible, and compatible with delicate 3D tissue, especially where viral prep time or cargo size is limiting.Typical advantages labs cite:
- Precise pulse control to balance delivery and viability
- Compatibility with large cargo (plasmids, mRNA, CRISPR RNPs)
- No viral production step
- Electrode format flexibility (cuvettes, chambers, tweezers) for different organoid sizes/types
- Rapid iteration (great for pilot testing)
- Support for mosaic phenotypes and lumen-targeted delivery
Method snapshots
NEPA21 electroporation in brain organoids
When labs choose it
- Early neural induction/neuroepithelial cyst stages ( often day 15–30).
- Mosaic, cell-autonomous phenotypes (polarity, migration, differentiation)
- Large constructs or CRISPR RNP delivery
- Lumen targeting (“apical electroporation” analog to In Utero electroporation).
Typical strengths
- Fast setup; rapid go/no-go results
- Strong expression in lumen-facing progenitors (when access is good).
- No integration footprint
- Mosaicism supports within-organoid comparisons
Typical trade-offs
- Efficiency drops with organoid age and ECM thickness
- Transient expression; plasmids dilute with divisions
- Heterogeneous depth (apical >> deep neurons).
- Late-stage organoids can be fragile if parameters aren’t optimized.
Typical workflow
- Prepare early organoids (often pre-embed or briefly handled to allow access).
- Microinject DNA/RNP mix into lumen/rosette space.
- Apply NEPA21 poring + transfer pulses (optimize per stage/electrode/tissue.
- Recovery culture (often ROCK inhibitor) and re-embed if needed.
- Analyse over roughly 3–10 days for transient assays (or longer depending on readout).
Viral delivery (Lento or AAV)
When labs choose it
- Mature organoids (often around day 50–200 depending on model).
- Need stable labelling (GCaMP, synaptic reporters, tracing)
- Pooled CRISPRi/a or barcode lineage screens.
- Even labelling across multiple layers where feasible.
Typical strengths
- Gentle on tissue (no electroporation shock).
- Stable long-term expression (especially with lentivirus).
- Can select/sort infected cells (depending on design).
- AAV serotype choice can bias targeting by cell type/ layer.
Typical trade-offs
- Vector production/purchase adds time (often 1–2+ weeks).
- Size limits apply (AAV ~4.7 kb; lenti ~8–9 kb typical).
- Biosafety requirements (often BSL-2).
- MOI can influence transcriptional state; requires control.
Typical workflow
- Obtain titered viral stocks.
- Incubate intact organoids or slices in virus, (sometimes with agitation/spinoculation).
- Allow expression to develop (AAV often days; lentivirus often ≥7 days for stable readouts).
- Optional selection/sort for uniformity.
- Proceed to functional assays (imaging, electrophysiology, tracing, screens).
Stage-based rule of thumb
| Stage / model | Common choice | Why |
|---|---|---|
| Neuroepithelial cysts (day ~10–20) | NEPA21 | Lumen accessible; fast; IUE-style targeting |
| Early cortical plate-like (day ~30–60) | Either ( depends on size) | Smaller organoids tolerate NEPA21, viral helps uniformity/ stability |
| Mature cortical organoids (day > ~90) | Viral | Thick tissue; higher electroporation risk; long-term functional assays |
| Slice cultures/fused assembloids | Viral | More even spread across layers; regional targeting potential |
Electrode format guide for NEPA21
Cuvette
Best for upstream cell engineering, organoid pieces, or fragments.
Glass chamber
Best for microinject-then-electroporate workflows in whole organoids.
Tweezer electrodes
Best for localized delivery where clamping the tissue is practical.
Typical use cases you can map to your goal
| Goal | Common choice | Why |
|---|---|---|
| Quick promoter/ protein localization check | NEPA21 plasmid | Rapid transient readout |
| CRISPR/Cas9 RNP knockout in organoid | NEPA21 | Large cargo; mosaic knockout for cell-autonomous effects |
| Stable reporter (GCaMP, synapsin-GFP) | Lentivirus | Long-term expression for functional assays |
| Inducible CRISPRi/a or pooled screens | Lentivirus | Stable integration and selection |
| Connectivity mapping/tracing | AAV (serotype-specific) | Tropism + longer assay |
|
How to choose for your experiment Choose NEPA21 when your experiment is early-stage, lumen-accessible, cargo-heavy, or benefits from mosaic readouts. Choose viral delivery when your experiment depends on long-term stability, broader labelling, or mature tissue compatibility. For many labs, the most efficient path is not one or the other, but: |
Published examples in brain and cortical organoid-related workflows
Direct electroporation of cortical or brain organoids, 3D tissue:Examples where NEPA21 is used directly in cortical or brain organoids after microinjection or in chamber or cuvette formats.
Taniguchi-Ikeda et al., iScience (2021)
Restoration of the defect in radial glial fiber migration and cortical plate organization in a brain organoid model of Fukuyama muscular dystrophy.FCMD brain organoid migration assay
Delivered: plasmid.
Model/stage: cortical organoids with microinjection into ventricle or rosette regions.
Hardware: electroporation glass chamber filled with Opti-MEM and plate electrodes in the chamber.
NEPA21 use-point: microinject then whole-organoid electroporation.
Notable detail: reported pulse conditions were 5 pulses, 125 V, 50 ms, with 1 second intervals.
Buijsen et al., Biomedicines (2024)
Calcium-enhanced medium-based delivery of splice-modulating antisense oligonucleotides in 2D and 3D hiPSC-derived neuronal models.ASO delivery in 2D/3D hiPSC-derived neuronal models.
Delivered: antisense oligonucleotides (ASOs).
Model/stage: hiPSC-derived cortical organoids.
Hardware: electroporation cuvette placed into a CU500 cuvette chamber.
NEPA21 use-point: organoid electroporation in cuvette format.
Hendriks et al., Cell (2024)
Human fetal brain self-organizes into long-term expanding organoids.Long-term expanding fetal brain organoids
Delivered: not listed here (method focus is the platform)
Model/stage: fetal brain organoid pieces
Hardware: 2 mm gap cuvette
NEPA21 use-point: whole-organoid or organoid-piece electroporation in cuvette format
Yamauchi et al., bioRxiv (posted 2025)
Heterochronic scaling of neurogenesis for species-specific dosing of cortical excitatory subtypes.Cortex-related workflow.
Hardware: CUY650P5 tweezer electrodes with 5 mm platinum disk.
NEPA21 use-point: tissue-level electroporation with tweezer format.
Examples where NEPA21 is used to edit hPSCs or iPSCs first, followed by organoid generation.
Pagliaro et al., Nature Communications (2023)
Temporal morphogen gradient-driven neural induction shapes single expanded neuroepithelium brain organoids with enhanced cortical identity.What they did: electroporated hESCs used downstream for cortical organoid work.
Hardware: 2 mm gap cuvettes, EC-002S.
NEPA21 use-point: upstream cell engineering step.
Kim et al., Molecular Cells (2022)
Aberrant cortical layer development of brain organoids derived from Noonan syndrome iPSCs.What they did: gene-editing step for iPSCs used to generate cortical organoids.
Hardware: cuvettes with NEPA21, gap or model not specified.
NEPA21 use-point: upstream cell editing.
Hong et al., Bioengineering & Translational Medicine (2024)
AAVS1-targeted stable expression of ChR2 in human brain organoids for consistent optogenetic control.AAVS1-targeted stable ChR2 expression for brain organoids
What they did: engineered hPSCs for forebrain organoids.
Hardware: 1 mm gap cuvette and CUY650P5 listed alongside NEPA21.
NEPA21 use-point: upstream engineering for consistent downstream optogenetics.
Kim et al., Science Advances (2025)
Perturbed cell fate decision by schizophrenia-associated AS3MTd2d3 isoform during corticogenesis.Corticogenesis/schizophrenia-associated isoform study
As listed: NEPA21 plus 2 mm gap cuvettes, EC-002S.
NEPA21 use-point: upstream engineering supporting downstream organoid experiments.
Talk to us about your organoid workflow
Share your organoid stage or size, cargo type, desired expression pattern, and readout timeline.
We can help recommend:
- the best delivery approach
- the most suitable electrode format
- a practical optimization strategy aligned to your assay