Electroporation-mediated gene transfer system applied to cultured CNS neurons
Electroporation-mediated gene transfer system applied to cultured CNS neurons
NEPA21 / CUY21 Applications . |
|
[Brain Slice] Electroporation-mediated gene transfer system applied to cultured CNS neurons . |
|
. . |
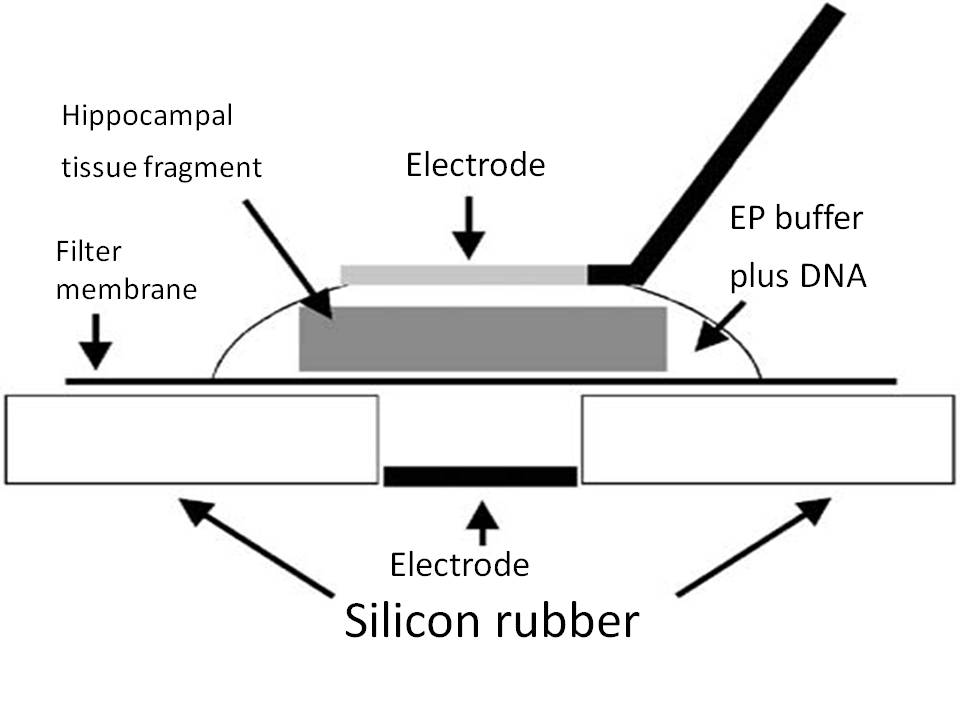
Schematic representation of an electroporation set-up. A fragment of the mouse embryonic hippocampus was placed on a Millipore membrane filter and 5µl EP buffer containing 1mg/ml of plasmid DNA was applied onto the tissue. A tungsten needle was attached to the surface of a droplet. After application of square pulses the tissue fragment was returned to a petri dish containing ice-cold HBSS solution. |
 |
Electroporation-mediated expression of fluorescent proteins in hippocampal neurons. (a-c) Organ culture of hippocampal tissue fragments three days after electroporation with CAG-eGFP (a), T∞1X4 -eGFP (b), and ß-actin-eGFP (c) expression constructs. (d, e) A mature hippocampal neuron maintained 14 days in dissociated culture after electroporation of a ß-actin-eGFP expression construct. Higher magni¢cation view of the region marked by a rectangle in (d) reveals dendritic spines on the surface of dendritic shafts (arrows in e). (f, g) A hippocampal neuron 7 days after electroporation of 1:1 mixture of T∞1X4-eGFP and T∞1X4-mRFP1. Both eGFP fluorescence (f) and mRFP1 fluorescence (g) can be observed in a single cell. (h) Relative fluorescence intensity of hippocampal tissue fragments after electroporation of eGFP-expression plasmids with four different promoter sequences. The tissue fragments were maintained in culture for 4 days, fixed and observed under a confocal microscope. Fluorescence intensities per unit area of the tissue fragments were determined. (i) Relative fluorescence intensity of hippocampal tissue fragments isolated at two different developmental stages and electroporated with ß-actin-eGFP. Tissue fragments were maintained for 4 days in culture and subsequently fixed. Fluorescence intensities were measured using a confocal microscope. Bars = 50µm (a-d, f, g); 10µm (e). |
|
Department of Anatomy and Cell Biology, School of Medicine, Tokyo Medical and Dental University; Shigeo Okabe *Neuroreport, Volume 15, Issue 6, Pages 971-975, April 29, 2004 |
|
| Contact Us
. |
|